How Soon After A Surgery To Repair Necrosis Can You Remove Tissue Expander And An Implant?
Risks of Chest Implants
Some of the complications and adverse outcomes of breast implants include:
- Implant complications, such as breast pain and changes in nipple and breast sensation
- Additional surgeries, with or without removal of the device (also see Implant Removal Options)
- Capsular contracture, scar tissue (sheathing) that forms effectually the implant and squeezes the implant
- Rupture and deflation
- Chest implant associated-anaplastic large cell lymphoma (BIA-ALCL), a blazon of non-Hodgkin's lymphoma (cancer of the immune system)
- Connective tissue affliction, breast cancer, and reproductive problems
- Systemic symptoms
- Breastfeeding
- Effects on children
Implant Complications
The post-obit is a list of local complications and adverse outcomes that occur in at least ane percentage of breast implant patients at any time. You may need non-surgical treatments or additional surgeries to care for any of these, and you should discuss whatever complication and necessary treatment with your doctor. These complications are listed alphabetically, not in club of how often they occur.
| Complication | Description |
|---|---|
| Asymmetry | The breasts are uneven in appearance in terms of size, shape or breast level. |
| Breast Pain | Pain in the nipple or chest expanse |
| Breast Tissue Cloudburst | Thinning and shrinking of the pare |
| Calcification/Calcium Deposits | Hard lumps under the pare around the implant. These can be mistaken for cancer during mammography, resulting in boosted surgery. |
| Capsular Contracture | Tightening of the tissue capsule effectually an implant, resulting in firmness or hardening of the chest and squeezing of the implant if severe. |
| Chest Wall Deformity | Chest wall or underlying rib muzzle appears deformed. |
| Deflation | Leakage of the saltwater (saline) solution from a saline-filled chest implant, often due to a valve leak or a tear or cutting in the implant shell (rupture), with fractional or complete collapse of the implant. |
| Delayed Wound Healing | Incision site fails to heal ordinarily or takes longer to heal. |
| Extrusion | The skin breaks down and the implant appears through the peel. |
| Hematoma | Collection of claret near the surgical site. May cause swelling, bruising and pain. Hematomas usually occur presently after surgery, but can occur any time at that place is injury to the chest. The body may absorb small hematomas, only large ones may require medical intervention, such as surgical draining. |
| Iatrogenic Injury/Impairment | Injury or harm to tissue or implant as a effect of implant surgery |
| Infection, including Toxic Shock Syndrome | Occurs when wounds are contaminated with microorganisms, such every bit leaner or fungi. Most infections resulting from surgery appear within a few days to a week, but infection is possible whatever fourth dimension subsequently surgery. If an infection does not respond to antibiotics, the implant may need to be removed |
| Inflammation/Irritation | Response by the torso to an infection or injury. Demonstrated by redness, swelling, warmth, pain and or/loss of role. |
| Lymphedema or Lymphadenopathy | Swollen or enlarged lymph nodes |
| Malposition/Displacement | The implant is not in the correct position in the breast. This can happen during surgery or afterwards if the implant moves or shifts from its original location. Shifting tin can exist acquired by factors such as gravity, trauma or capsular contracture. |
| Necrosis | Dead pare or tissue effectually the breast. Necrosis tin be caused past infection, use of steroids in the surgical breast pocket, smoking, chemotherapy/radiation, and excessive heat or cold therapy. |
| Nipple/Breast Awareness Changes | An increase or decrease in the feeling in the nipple and/or breast. Can vary in degree and may be temporary or permanent. May bear on sexual response or breast feeding. |
| Palpability | The implant tin can be felt through the skin. |
| Ptosis | Breast sagging that is ordinarily the outcome of normal aging, pregnancy or weight loss. |
| Redness/Bruising | Haemorrhage at the time of surgery tin cause the skin to change colour. This is an expected symptom due to surgery, and is likely temporary. |
| Rupture | A tear or hole in the implant'south outer crush. |
| Seroma | Drove of fluid around the implant. May crusade swelling, pain and bruising. The body may absorb small seromas. Large ones will crave a surgical bleed. |
| Pare Rash | A rash on or around the breast. |
| Unsatisfactory Style/Size | Patient or doctor is non satisfied with the overall look based on the way or size of the implant used. |
| Visibility | The implant can be seen through the skin. |
| Wrinkling/Rippling | Wrinkling of the implant that can be felt or seen through the skin. |
A complete list of complications, also every bit information on rates for those complications tin can be found in the patient labeling for the approved breast implants, Labeling for Approved Breast Implants.
Boosted Surgeries
Breast Implants are not considered lifetime devices. The longer people accept them, the greater the chances are that they volition develop complications, some of which volition require more surgery. At that place is no guarantee that you will have a satisfactory cosmetic result from any reoperation.
The type of surgical process performed during a reoperation depends on the complication involved. You may need to have one or more than reoperations over the course of your life due to one complexity or a combination of local complications. More than one process may be performed in a unmarried reoperation. Types of surgical procedures that may exist performed in a reoperation include:
- Implant removal, with or without replacement
- Capsule removal or surgical release of the scar tissue around the breast implant
- Scar or wound revision, such as surgical removal of excess scar tissue
- Drainage of a hematoma by inserting a needle or tube through the skin to bleed the collection of claret
- Repositioning of the implant by surgically opening the incision and moving the implant
- Biopsy/cyst removal by inserting a needle through the peel or cut through the skin to remove a lump.
Implant Removal Options
If you elect to accept your chest implants removed, or if information technology is medically indicated, there are two chief methods for implant removal. Your plastic surgeon may choose to remove your implant alone and leave the scar tissue that surrounds your implant in your body, also called the scar sheathing. This option requires less surgical dissection and may pose less take chances of local complications such as bleeding. Alternatively, your surgeon may also surgically remove the scar capsule when your breast implant is removed. This is called "en-bloc resection". You should discuss with your surgeon which method is best for your situation.
If you feel whatever symptoms of BIA-ALCL, such as persistent swelling or pain, or other changes in the area effectually your breast implant, talk to your surgeon or health care provider almost the need for further evaluation. Evaluation for BIA-ALCL typically involves a physical test, imaging, and/or assessment of the fluid or tissue around the chest implant. It is important to have an evaluation to diagnose BIA-ALCL because a confirmed BIA-ALCL diagnosis may modify the type of functioning that should be performed.
Generally, patients with confirmed BIA-ALCL should undergo implant removal and removal of the surrounding scar capsule, which is a more than extensive operation than implant removal solitary. Talk to your surgeon most the method of removal most appropriate for yous.
Removal of a breast implant, with or without replacement, is one type of reoperation. The life of breast implants varies by person and can't be predicted. You may need to have your implant removed at some time over the form of your life because of one or more than local complications.
After removal, some women practise not choose to replace their implants. These women may have cosmetically undesirable dimpling, chest wall concavity, puckering, or sagging of their natural breasts.
The photo below shows a 29-year-old woman 1 year after having her silicone gel-filled breast implants removed, but not replaced. Women with big breast implants, especially those inserted on top of the chest muscles (subglandularly), may take major cosmetic deformity if they choose not to supplant them or to undergo additional reconstructive surgery.
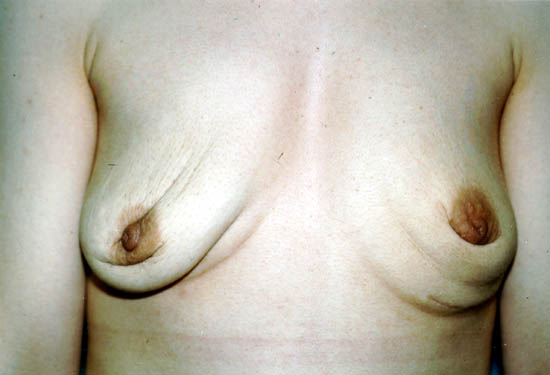
Photo courtesy of Walter Peters, Ph.D., M.D., F.R.C.Southward.C., University of Toronto.
Some insurance companies do not cover implant removal or implant replacement, fifty-fifty if there are complications and fifty-fifty if the first implant surgery was covered.
Capsular Contracture
Capsular contracture is the hardening of the chest around the implant. Information technology tin occur in the tissue surrounding one or both implants. This hardening causes the tissue to tighten, which tin be painful.
Capsular contracture may be more common post-obit infection, hematoma and seroma. Nonetheless, the cause of capsular contracture is not known.
There are four grades of capsular contracture, known as Baker grades.
Baker Grading Scale
- Grade I: Chest is unremarkably soft and looks natural
- Grade II: Breast is a lilliputian firm but looks normal
- Grade III: Breast is firm and looks abnormal
- Class IV: Breast is hard, painful, and looks abnormal
Grades III and 4 capsular contracture are considered severe and may require reoperation. The surgical procedure usually involves removal of the implant with or without replacement of the implant. In that location is a possibility that capsular contracture could occur again after surgery to right it.
The FDA has not cleared or approved whatever devices to treat or reduce the incidence of capsular contracture.
The moving picture below shows a Class IV capsular contracture in the right breast of a 29-year-old woman 7 years after placement of silicone gel-filled breast implants.
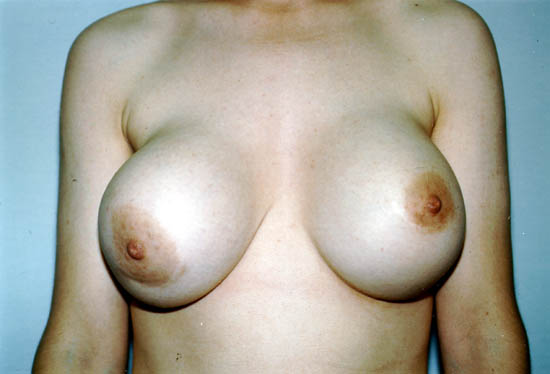
Photo courtesy of Walter Peters, Ph.D., Thou.D., F.R.C.S.C., University of Toronto.
Rupture and Deflation
Rupture is a tear or hole in the outer beat of the breast implant.
Some possible causes of rupture of breast implants include:
- Capsular contracture
- Pinch during a mammogram
- Damage past surgical instruments
- Damage during procedures to the breast, such as biopsies and fluid drainage
- Normal crumbling of the implant
- Overfilling or underfilling of saline-filled chest implants
- Physical stresses such as trauma or intense physical pressure
- Placement through a non-FDA approved incision site, for case the belly push
- Too much handling during surgery

Saline-Filled Breast Implant Rupture and Deflation
The term rupture is used for all types of chest implants, merely the term deflation is simply used for saline-filled implants. You lot and/or your doctor will exist able to tell if your saline-filled implant ruptures because the saline solution leaks into your body immediately or over several days and deflates the implant. You volition find that your implant loses its original size or shape.
The following surgical procedures are non recommended for FDA-approved saline-filled breast implants considering they are known to crusade rupture and deflation:
- Airtight capsulotomy - a technique used to salve capsular contracture involving manually squeezing the breast to break the difficult sheathing
- Placement of drugs or other substances inside the implant other than sterile saline
- Any contact of the implant with Betadine, a povidone-iodine topical antiseptic made by Purdue Frederick Company
- Injection through the implant beat
- Alteration of the implant
- Stacking of the implants (more than ane implant per breast pocket).
The photograph below shows a xxx-yr-old woman whose left saline-filled breast implant deflated. The implant is thought to have deflated due to a particular design, which is no longer used by the manufacturer.
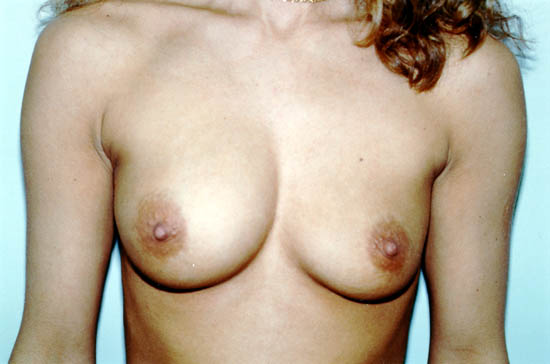
Photo courtesy of Walter Peters, Ph.D., G.D., F.R.C.Due south.C., Academy of Toronto.
Silicone Gel-Filled Implant Rupture
Silicone breast implants can rupture at whatever fourth dimension after your implant surgery, but the longer an implant is in place, the greater the possibility an implant may rupture.
If a silicone gel-filled breast implant ruptures, it is non likely that you or your doc will immediately notice because most silicone implant ruptures are without symptoms, "silent ruptures". A silent rupture doesn't alter the way an implant looks or feels, and your surgeon or health care provider may not be able to detect a silent rupture past a concrete examination solitary. Magnetic resonance imaging (MRI) is the most effective method for detecting silent rupture of silicone gel-filled breast implants.
However, occasionally when a silicone gel-filled implant ruptures, a woman may notice a decrease in breast size, alter in breast implant shape, difficult lumps over the implant or chest area, an uneven appearance of the breasts, pain or tenderness, tingling, swelling, numbness, burning, or changes in sensation.
Generally, when silicone gel-filled implants rupture, the silicone gel escapes through a tear or hole in the implant shell but remains confined inside the scar tissue capsule effectually the implant, called an intra-capsular rupture. If the gel migrates beyond the scar tissue effectually the chest implant, it is chosen an extracapsular rupture. Sometimes, after a rupture, the gel may motion to other distant areas around the body, which is called extracapsular rupture with gel migration. It may be difficult to remove silicone gel later on a rupture.
Connective Tissue Illness, Breast Cancer, and Reproductive Issues
The FDA has non detected any association between silicone gel-filled breast implants and connective tissue disease, breast cancer, or reproductive problems. Still, the FDA has received reports of systemic symptoms (run into beneath) by some patients with both saline and silicone gel-filled chest implants. In order to fully understand these complications, studies would demand to exist larger and longer than those conducted and so far.
Systemic Symptoms [Breast Implant Disease (BII)]
Symptoms such as fatigue, retention loss, rash, "brain fog," and joint hurting may be associated with breast implants. Some patients may utilise the term "chest implant illness" (BII) to describe these symptoms. Researchers are investigating these symptoms to amend understand their origins. These symptoms and what causes them are poorly understood. In some cases, removal of the breast implants without replacement is reported to reverse symptoms of breast implant disease.
We encourage patients to report any injury, adverse event, or symptom related to a medical device, including the symptoms listed above, to the FDA past phone at ane-800-FDA-1088 or online at MedWatch, the FDA Prophylactic Data and Adverse Consequence Reporting program. Please include the post-obit data:
- Device Name (Brand Proper noun)
- Manufacturer's Name
- Details of Agin Event and Medical and/or Surgical Interventions (if applicable)
Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL)
Chest implant-associated anaplastic large cell lymphoma (BIA-ALCL) is a T-jail cell lymphoma that can develop following breast implants. For additional data, see: Breast Implant-Associated Anaplastic Big Cell Lymphoma (BIA-ALCL).
Breastfeeding
Some women who undergo breast augmentation can successfully breastfeed and some cannot. Women who undergo mastectomies and and then have breast implant reconstruction surgeries may not exist able to breastfeed on the afflicted side due to loss of breast tissue and the glands that produce milk.
Effects on Children
At this time, it is not known if a small amount of silicone may pass through from the breast implant silicone shell into breast milk during breastfeeding. Although at that place are currently no established methods for accurately detecting silicone levels in chest milk, a written report measuring silicon (one component in silicone) levels did non point college levels in breast milk from women with silicone gel-filled implants when compared to women without implants.
In addition, concerns have been raised regarding potential damaging effects on children born to mothers with implants. Ii studies in humans take found no increased risk of birth defects in children born to mothers who have had breast implant surgery. Although low birth weight was reported in a third study, other factors (for example, lower pre-pregnancy weight) may explain this finding.
Source: https://www.fda.gov/medical-devices/breast-implants/risks-and-complications-breast-implants
Posted by: shuttleworthmillan91.blogspot.com


0 Response to "How Soon After A Surgery To Repair Necrosis Can You Remove Tissue Expander And An Implant?"
Post a Comment